 I hope it’s clear now that dS=dQ/(interface contact temp) +dSp. I found it easy to get terms like ‘entropy production’ (dSp = dSgen) and ‘entropy increase’ (dS) mixed up.įor a long time, I was also confused by textbooks which said things like “dSgen includes the effects of finite T differences.” So, when you put an object at say 2T in contact with a system at T, did that mean that, in dS=dQ/T +dSgen, the term dQ/T=0 (because the ht was finite-difference and thus irreversible and dSgen included all effects of irreversibility)? Also, what T value was I supposed to use? 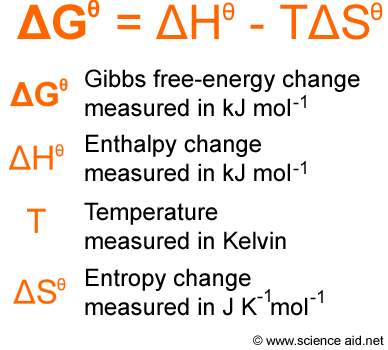
It’s quite important to keep a close eye on the language used. It might include the effects of internal friction, pressure gradients etc…anything which represents a deviation from quasi-equilibrium in the system. This is a term the detailed behaviour of which is hard to account for. The extra increase in system entropy which is created, after the effect of transfer of heat in (dQ/T), is summed up as dSgen (aka dSp). The subsequent passage of heat into the system is therefore irreversible (it’s very hard to keep track of which atoms hit which others and thus how the dissipation process plays out). Any such non uniformities result in non equilibrium conditions. Moreover, the entropy change is calculated by using the relative chemical potential that is defined based on the equilibrium state and not on the standard chemical potential. In reality, once the dQ has entered the system, it is faced by some kind of temperature profile (ie the internal T distribution in the system is non-uniform). Then the entropy production is applied for searching the parameter region where the pattern is bistable. Thus the change in the internal energy of the system is related to the change in entropy, the absolute temperature, and the PV work done. wrev PV, we can express Equation 13.4.3 as follows: U qrev + wrev TS PV. That would be the ideal case of reversible heat transfer (dS=dQ/T). Recognizing that the work done in a reversible process at constant pressure is. Now it may be that the rest of the system is also at T, in which case there are no thermal gradients and no additional changes in system entropy. 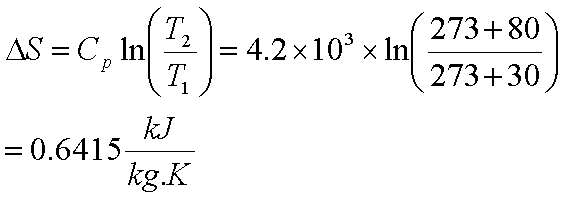 
Perhaps the single most important thing to remember, however, is that, in the equation for dS, when you transfer heat (dQ) into a system, the local system temperature at the interface is what is meant by T. This is clearly not true, since we can (in theory) increase the entropy of a system just by transferring some heat to it reversibly. The first confusion I suffered from was the idea that entropy increase was always a sign of irreversibility. This is one of those parts of engineering thermodynamics courses which is never made clear enough. The equation for the change in entropy, S, is. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
